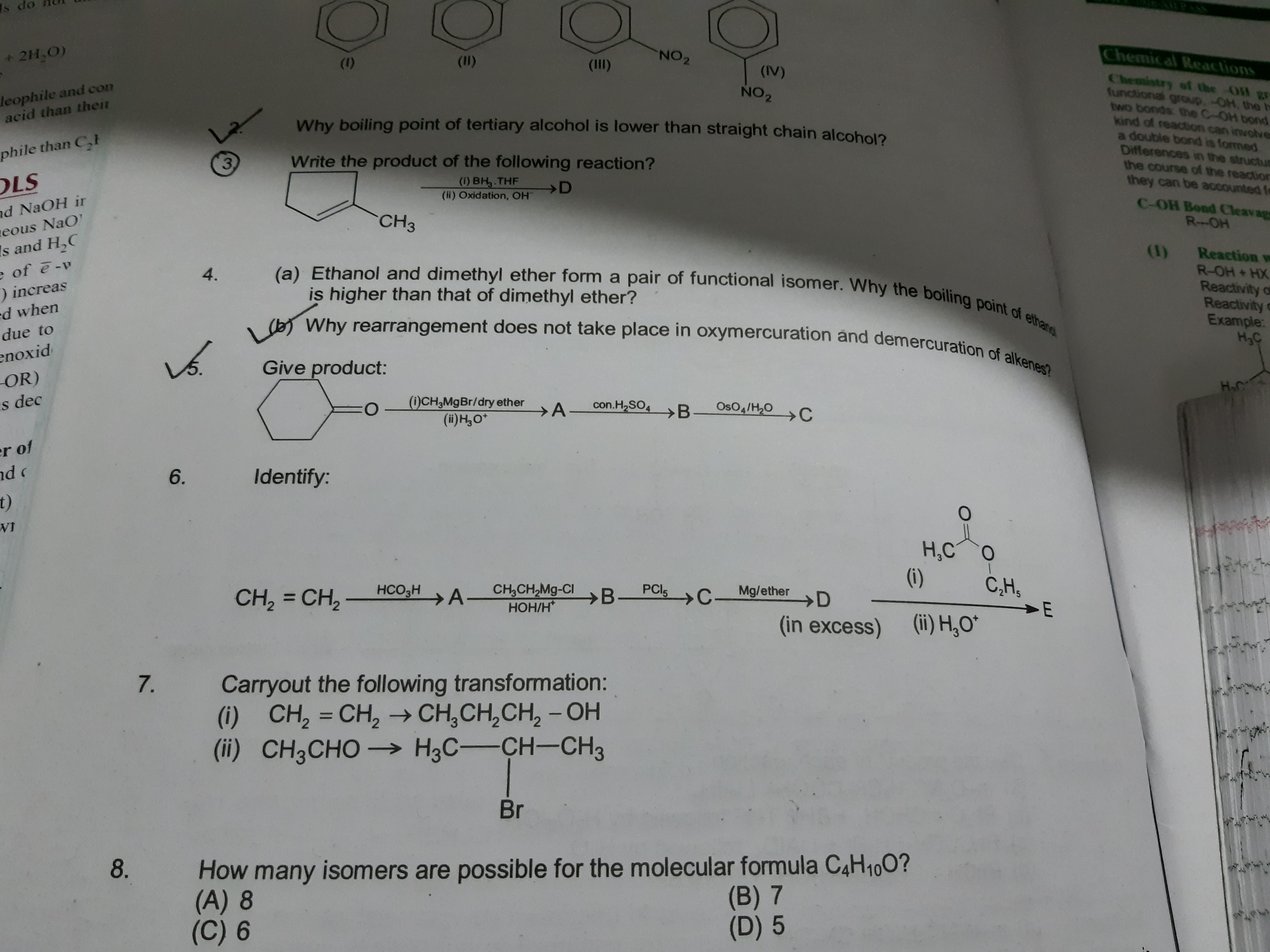
In these cases, the – e ending of the parent alkane is retained.įigure 14.1 “IUPAC Rules for Alcohols” shows some examples of the application of these rules. If more than one OH group appears in the same molecule (polyhydroxy alcohols), suffixes such as – diol and – triol are used.(In cyclic alcohols, the carbon atom bearing the OH group is designated C1, but the 1 is not used in the name.) Substituents are named and numbered as in alkanes. The number that indicates the position of the OH group is prefixed to the name of the parent hydrocarbon, and the – e ending of the parent alkane is replaced by the suffix – ol.The chain is numbered from the end nearest the OH group. As noted in Chapter 4 Covalent Bonding and Simple Molecular Compounds, an alcohol is an organic compound with a hydroxyl (OH) functional group on an aliphatic carbon atom. Show Answer Key Takeaways Alcohols can be dehydrated to form either alkenes (higher temperature, excess acid) or ethers (lower temperature, excess alcohol). Is Y oxidized, reduced, or neither Explain. The longest continuous chain (LCC) of carbon atoms containing carbon atom to which the the OH group is attached is considered the parent compound-an alkane with the same number of carbon atoms. In a reaction, 2 mol of compound Y with the molecular formula C 4 H 10 O is converted to 1 mol of compound Z with the formula C 8 H 18 O.The R group tool is located in the charges and lone pairs drop-down menu. 
Here are some basic IUPAC rules for naming alcohols: Waference Draw general formulas for an alcohol and a phenol, showing the functional group: Represent the nonaromatic hydrocarbon group as R1. They are organic derivatives of water where a hydrogen ion is replaced by an alkyl group. The family also includes such familiar substances as cholesterol and the carbohydrates.Īs we noted in Chapter 4 “Covalent Bonding and Simple Molecular Compounds”, Section 4.6 “Introduction to Organic Chemistry”, methanol (CH 3OH) and ethanol (CH 3CH 2OH) are the first two members of the homologous series of alcohols.Īccording to the International Union of Pure and Applied Chemistry (IUPAC), alcohols are named by changing the ending of the parent alkane name ( Chapter 12 “Organic Chemistry: Alkanes and Halogenated Hydrocarbons”, Section 12.5 “IUPAC Nomenclature”) to – ol. Chemical Compound: Alcohols General Formula and Functional Group Alcohols are functional groups that are characterized by one or more OH group attached to a hydrocarbon chain. Most people are familiar with ethyl alcohol (ethanol), the active ingredient in alcoholic beverages, but this compound is only one of a family of organic compounds known as alcohols. Table 12.4 “Common Alkyl Groups” presents some common alkyl groups.)Īlcohols are common in nature. (For more information about alkyl groups, see Chapter 12 “Organic Chemistry: Alkanes and Halogenated Hydrocarbons”, Section 12.5 “IUPAC Nomenclature”. 2D Graphics The best graphics in chemistry Pixel Perfect We spend a very long time scrutinizing the graphics output in ChemDoodle. Because OH is the functional group of all alcohols, we often represent alcohols by the general formula ROH, where R is an alkyl group. ChemDoodle 2D contains thousands of chemistry features, helping you produce the highest quality graphics and saving you hours of work.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
